It is used in making LEDs (light-emitting diodes).Phosphorus is used in the manufacturing of bronze and steel.Phosphorus is used in making of safety matches, and various ammunitions, such as incendiary shells and hand grenades etc.
It is an essential nutrient for the growth of plants. The largest use of phosphorus is in the production of fertilizers.Diphosphine (P2H4), is an analogue of hydrazine and is highly flammable. Phosphine (PH3) is a toxic compound with pungent smell, is structural analogue of ammonia.
There is a vast variety of phosphorus compounds including oxoacids (phosphoric acid), sulfides, nitrides (phosphorus nitride halogens (F2PN, Cl2PN), and phosphides (reaction of metals with red phosphorus). The most abundant compounds of phosphorus contain the tetrahedral anion of phosphate (PO 4 3-). In the presence of oxygen, white phosphorus emits a light green glow, termed as chemiluminescence (glow caused by cold chemical reaction). White phosphorus is very reactive element. It resembles graphite in appearance and structure. Black phosphorus is also termed as violet phosphorus and is the least reactive allotrope of phosphorus. White phosphorus is a highly toxic substance., while red phosphorus is non-toxic. Red phosphorus (which is formed by heating of white phosphorus at high temperature) ignites on friction. There are two allotropic forms of phosphorus, red and black, that differ in physical and chemical properties. Phosphorus undergoes spontaneous ignition in air and forms pentoxide (P 4O 10). White phosphorus is yellowish white solid, that has a waxy texture.

The annual production of elemental phosphorus is around 1000,000 tons. Other countries or regions that are significant producers of phosphorus include Russia, China, Florida and Morocco. The largest natural reserves of phosphorus are present in Arab region. It is the 6 th most abundant element in the living systems. Phosphorus is an element of great biological value. The most common source of phosphorus is calcium phosphate that is present in rocks. It forms various compounds and is present in large variety of minerals. Phosphorus does not exist in free or elemental state in nature. Phosphorus Periodic Table ClassificationĬolourless, waxy white, yellow, scarlet, red, violet, black Later in 1850s, the presence of phosphate in rocks on the form of calcium phosphate was discovered and the phosphorus production switched form bones to rocks. Until 1840s, one of the major sources of phosphorus has been ashes of bones, and until 1840s.

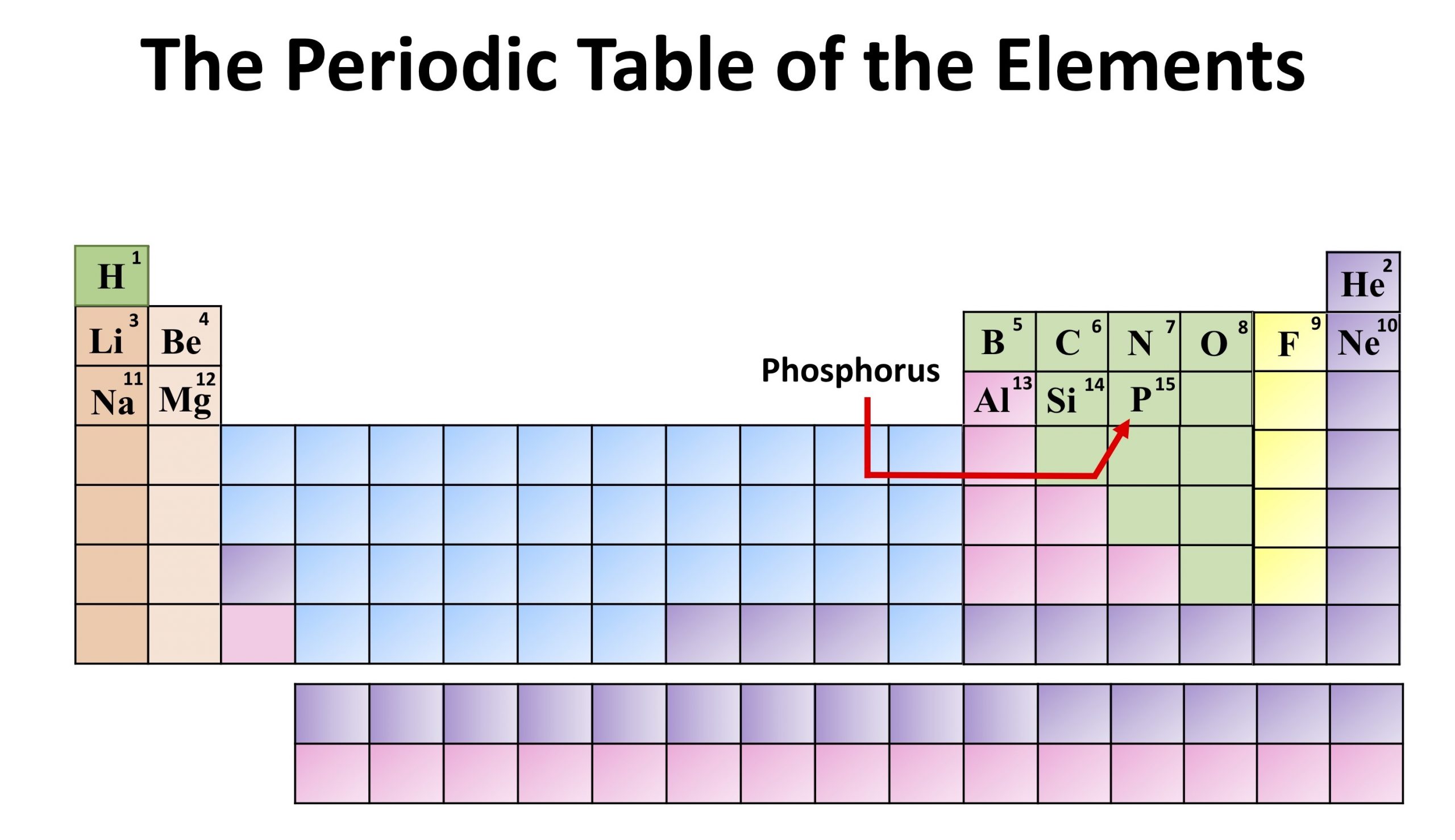
Gottlieb Gahn and Carl Wilhelm (1796) were the first to discover the presence of phosphorus, in the form of calcium phosphate, in the bones. Phosphorus was initially used on a wide scale to make poisons, explosives and nerve agents and due to its toxic and lethal properties, it was commonly named as the “devil’s element”. In 1680, Boyle introduced the use of phosphorus to ignite wooden splints with tips of sulfur, and these later were developed into matches. Commercial scale production of phosphorus was started by Brand and later many scientists, including Robert Boyle, used the same method of phosphorus production developed by Brand. The name phosphorus also has an interesting origin, as Phosphorus is the name of planet Venus in Ancient Greece language and it means “carrier of light” or “light-bringer”. Antoine Lavoisier identified phosphorus as a separate element in 1777. He obtained a white material that burned brilliantly and emitted a glow in the dark when exposed to oxygen. The discovery of phosphorus as a novel element was carried out by in Hennig Brand in 1669 by evaporating urine. It was discovered in 1669 by Hennig Brand and has two allotropic forms and eighteen isotopes. Phosphorus is a non-metal with diverse biological and industrial significance.

